All in One View
Content from Introducing the Shell
Last updated on 2026-02-20 | Edit this page
Overview
Questions
- What is a command shell and why would I use one?
- How can I move around in a computer?
- How can I see what files and directories I have?
- How can I specify the location of a file or directory on my computer?
Objectives
- Describe key reasons for learning shell.
- Learn how to access a remote machine.
- Navigate your file system using the command line.
- Access and read help files for
bashprograms and use help files to identify useful command options. - Demonstrate the use of tab completion, and explain its advantages.
What is a shell and why should I care?
A shell is a computer program that presents a command line interface which allows you to control your computer using commands entered with a keyboard instead of controlling graphical user interfaces (GUIs) with a mouse/keyboard combination.
There are many reasons to learn about the shell.
- Many bioinformatics tools can only be used through a command line interface, or have extra capabilities in the command line version that are not available in the GUI. This is true, for example, of BLAST, which offers many advanced functions only accessible to users who know how to use a shell.
- The shell makes your work less boring. In bioinformatics you often need to do the same set of tasks with a large number of files. Learning the shell will allow you to automate those repetitive tasks and leave you free to do more exciting things.
- The shell makes your work less error-prone. When humans do the same thing a hundred different times (or even ten times), they’re likely to make a mistake. Your computer can do the same thing a thousand times with no mistakes.
- The shell makes your work more reproducible. When you carry out your work in the command-line (rather than a GUI), your computer keeps a record of every step that you’ve carried out, which you can use to re-do your work when you need to. It also gives you a way to communicate unambiguously what you’ve done, so that others can check your work or apply your process to new data.
- Many bioinformatic tasks require large amounts of computing power and can’t realistically be run on your own machine. These tasks are best performed using remote computers or cloud computing, which can only be accessed through a shell.
In this lesson you will learn how to use the command line interface to move around in your file system.
How to access the shell
On a Mac or Linux computer, you can access a shell through a program called Terminal, which is already available on your computer. If you’re using Windows, you’ll need to download a separate program to access the shell (see installation instructions here). If you have all the bioinformatic programs installed in your local machine you are all set.
In this workshop, we suggest using a remote server to invest most of our time learning the basics of shell by manipulating some experimental data. The remote server already includes the required bioinformatics packages as well as the large datasets that usually take a lot of time to load into everyone’s local computers. If you are in a workshop from UNAM-CCM you will access the Bash shell and the Python notebook through a JupyterHub server. If you are in another workshop you will access them through an AWS remote machine.
Connection to the UNAM-CCM JupyterHub
Open the JupyterHub server login site in a new tab with this link.
Open this Google
sheet in a new tab and write your name in a user.
Use this user information to log in to the JupyterHub site that you
opened in the previous step.
To open a Bash Terminal click on the button “New” (upper right within
the “Files” section) and choose the option “Terminal” from the drop-down
menu. A new tab with a terminal will open.
Connection to the AWS remote machine
The instructor of your workshop will give you an
ip_address and password to login.
To log in you need to open the Terminal program and use the
ssh command (ssh stands for Secure Shell), your username
and the address of the machine you are logging into.
Then you are prompted to type the password. Take into account that while you are typing a password no characters will appear on the screen, trust that they are being typed and press enter.
After logging in, you will see a screen showing something like this:
OUTPUT
Welcome to Ubuntu 14.04.3 LTS (GNU/Linux 3.13.0-48-generic x86_64)
* Documentation: https://help.ubuntu.com/
System information as of Sat Feb 2 00:08:17 UTC 2019
System load: 0.0 Memory usage: 5% Processes: 82
Usage of /: 29.9% of 98.30GB Swap usage: 0% Users logged in: 0
Graph this data and manage this system at:
https://landscape.canonical.com/
Get cloud support with Ubuntu Advantage Cloud Guest:
http://www.ubuntu.com/business/services/cloud
597 packages can be updated.
444 updates are security updates.
New release '16.04.5 LTS' available.
Run 'do-release-upgrade' to upgrade to it.
Last login: Fri Feb 1 22:34:53 2019 from c-73-116-43-163.hsd1.ca.comcast.netThis provides a lot of information about the remote server that
you’re logging in to. We’re not going to use most of this information
for our workshop, so you can clear your screen using the
clear command.
This will scroll your screen down to give you a fresh screen and will make it easier to read. You haven’t lost any of the information on your screen. If you scroll up, you can see everything that has been output to your screen up until this point.
Navigating your file system
Prepare your genome database
Make sure you have the pan_workshop/ directory. If you
do not have it, you can download it with the following instructions.
The part of the operating system responsible for managing files and directories is called the file system. It organizes our data into files, which hold information, and directories (also called “folders”), which hold files or other directories.
Several commands are frequently used to create, inspect, rename, and delete files and directories.
Preparation Magic
If you type the command: PS1='\W\$ ' into your shell,
followed by pressing the Enter key, your window should look
like this:~\$
That only shows the ultimate directory where you ar standing. In this
case it is the home directory. The symbol ~ is an
abbreviation of the home directory. This isn’t necessary to follow along
(in fact, your prompt may have other helpful information you want to
know about). This is up to you!
The dollar sign is a prompt, which shows us that the shell is waiting for input; your shell may use a different character as a prompt and may add information before the prompt. When typing commands, either from these lessons or from other sources, do not type the prompt, only the commands that follow it. In this lesson we will use the dollar sign to indicate the prompt.
Let’s find out where we are by running a command called
pwd (which stands for “print working directory”). At any
moment, our current working directory is our current
default directory, i.e., the directory that the computer assumes we want
to run commands in unless we explicitly specify something else. Here,
the computer’s response is /home/dcuser, which is the top
level directory within our cloud system:
OUTPUT
/home/dcuserLet’s look at how our file system is organized. We can see what files
and subdirectories are in this directory by running ls,
which stands for “listing”:
OUTPUT
pan_workshop ls prints the names of the files and directories in the
current directory in alphabetical order, arranged neatly into columns.
We’ll be working within the pan_workshop subdirectory, and
creating new subdirectories, throughout this workshop.
The command to change locations in our file system is cd
followed by a directory name to change our working directory.
cd stands for “change directory”.
Let’s say we want to navigate to the pan_workshop
directory we saw above. We can use the following command to get
there:
Let’s look at what is in this directory:
OUTPUT
data We can make the ls output more comprehensible by using
the flag -F, which tells ls
to add a trailing / to the names of directories:
OUTPUT
data/ Anything with a “/” after it is a directory. Things with a “*” after them are programs. If there are no decorations, it’s a file.
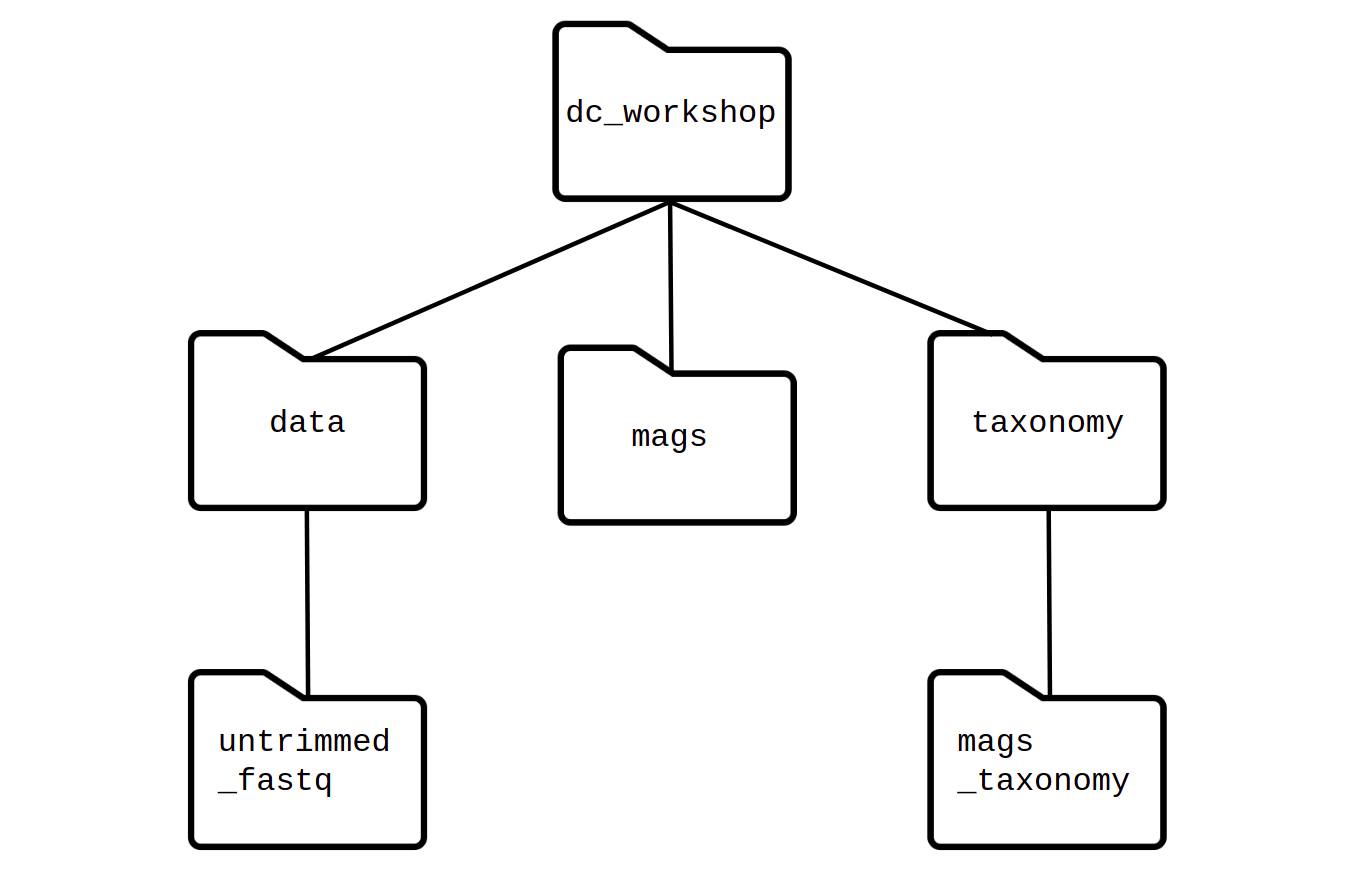
To understand a little better how to move between folders, let’s see the following image:
Here we can see a diagram of how the folders are arranged one inside
another. In this way, if we think about moving, from
pan_workshop to the data folder, the path must
go as they are ordered: cd pan_workshop/data
ls has lots of other options. To find out what they are,
we can type:
Some manual files are very long. You can scroll through the file using your keyboard’s down arrow or use the Space key to go forward one page and the b key to go backwards one page. When you are done reading, hit q to quit.
Exercise 1(Begginer): Extra information with
ls -l
Use the -l option for the ls command to
display more information for each item in the directory. What is one
piece of additional information this long format gives you that you
don’t see with the bare ls command?
No one can possibly learn all of these arguments, that’s why the manual page is for. You can (and should) refer to the manual page or other help files as needed.
Let’s go into the data/agalactiae_H36B directory and see
what is in there.
OUTPUT
Streptococcus_agalactiae_H36B.fna Streptococcus_agalactiae_H36B.gbkShortcut: Tab Completion
Usually the key Tab is located on the left side of the keyboard just above the “Shift” key or “Mayus” key.
Typing out file or directory names can waste a lot of time and it’s easy to make typing mistakes. Instead we can use tab complete as a shortcut. When you start typing out the name of a directory or file, then hit the Tab key, the shell will try to fill in the rest of the directory or file name.
Return to your home directory:
then enter:
The shell will fill in the rest of the directory name for
pan_workshop.
Now change directories to data in
pan_workshop
Using tab complete can be very helpful. However, it will only autocomplete a file or directory name if you’ve typed enough characters to provide a unique identifier for the file or directory you are trying to access.
The shell auto-completes your command to agalactiae_,
because there is another name in the directory begin with this prefix.
When you hit Tab again, the shell will list the possible
choices.
OUTPUT
agalactiae_18RS21/ agalactiae_H36B/Tab completion can also fill in the names of programs, which can be useful if you remember the beginning of a program name.
OUTPUT
pwd pwdxDisplays the name of every program that starts with
pw.
Summary
We now know how to move around our file system using the command line. This gives us an advantage over interacting with the file system through a Graphical User Interface (GUI) as it allows us to work on a remote server, carry out the same set of operations on a large number of files quickly, and opens up many opportunities for using bioinformatics software that is only available in command line versions.
In the next few episodes, we’ll be expanding on these skills and seeing how using the command line shell enables us to make our workflow more efficient and reproducible.
- The shell gives you the ability to work more efficiently by using keyboard commands rather than a GUI.
- Useful commands for navigating your file system include:
ls,pwd, andcd. - Most commands take options (flags) which begin with a
-. - Tab completion can reduce errors from mistyping and make work more efficient in the shell.
Content from Navigating Files and Directories
Last updated on 2026-02-20 | Edit this page
Overview
Questions
- How can I perform operations on files outside of my working directory?
- What are some navigational shortcuts I can use to make my work more efficient?
Objectives
- Use a single command to navigate multiple steps in your directory structure, including moving backwards (one level up).
- Perform operations on files in directories outside your working directory.
- Work with hidden directories and hidden files.
- Interconvert between absolute and relative paths.
- Employ navigational shortcuts to move around your file system.
Moving around the file system
We’ve learned how to use pwd to find our current
location within our file system. We’ve also learned how to use
cd to change locations and ls to list the
contents of a directory. Now we’re going to learn some additional
commands for moving around within our file system.
Use the commands we’ve learned so far to navigate to the
pan_workshop/data/ directory, if you’re not already
there.
What if we want to move back up and out of this directory and to our
top level directory? Can we type cd pan_workshop? Try it
and see what happens.
OUTPUT
-bash: cd: pan_workshop: No such file or directoryYour computer looked for a directory or file called
pan_workshop within the directory you were already in. It
didn’t know you wanted to look at a directory level above the one you
were located in.
We have a special command to tell the computer to move us back or up one directory level.
Now we can use pwd to make sure that we are in the
directory we intended to navigate to, and ls to check that
the contents of the directory are correct.
OUTPUT
/home/dcuser/pan_workshop/dataFrom this output, we can see that .. did indeed took us
back one level in our file system.
You can chain these together to move several levels:
First use the man command to look at the options for
ls.
The -a option is short for all and says
that it causes ls to “not ignore entries starting with .”
This is the option we want.
OUTPUT
. .. agalactiae_18RS21 agalactiae_H36B .hidden annotated_miniThe name of the hidden directory is .hidden. We can
navigate to that directory using cd.
And then list the contents of the directory using
ls.
OUTPUT
youfoundit.txtThe name of the text file is youfoundit.txt.
File permissions
Another option that the ls command has is to check the
permissions on a file. If we are organized and we have a folder with the
backup of all our files, we can rescue files that we have accidentally
deleted, for example, but just because we have two copies doesn’t make
us safe. We can still accidentally delete or overwrite both copies. To
make sure we can’t accidentally mess up a file, we’re going to change
the permissions on the file so that we’re only allowed to read
(i.e. view) the file, not write to it (i.e. make new changes).
View the current permissions on a file using the -l
(long) flag for the ls command.
OUTPUT
total 0
-rw-rw-r-- 1 dcuser dcuser 0 May 27 23:16 youfoundit.txtThe first part of the output for the -l flag gives you
information about the file’s current permissions. There are ten slots in
the permissions list. The first character in this list is related to
file type, not permissions, so we’ll ignore it for now. The next three
characters relate to the permissions that the file owner has, the next
three relate to the permissions for group members, and the final three
characters specify what other users outside of your group can do with
the file. We’re going to concentrate on the three positions that deal
with your permissions (as the file owner).
Here the three positions that relate to the file owner are
rw-. The r means that you have permission to
read the file, the w indicates that you have permission to
write to (i.e. make changes to) the file, and the third position is a
-, indicating that you don’t have permission to carry out
the ability encoded by that space (this is the space where
x or executable ability is stored, we’ll talk more about
this in a
later lesson).
Our goal for now is to change permissions on this file so that you no
longer have w or write permissions. We can do this using
the chmod (change mode) command and subtracting
(-) the write permission -w.
OUTPUT
total 0
-r--r--r-- 1 dcuser dcuser 0 May 27 23:16 youfoundit.txtAbsolute vs. relative paths
The cd command takes an argument which is a directory
name. Directories can be specified using either a relative path
or a full absolute path. The directories on the computer are
arranged into a hierarchy. The full path tells you where a directory is
in that hierarchy. Navigate to the home directory, then enter the
pwd command.
You will see:
OUTPUT
/home/dcuserThis is the full name of your home directory. This tells you that you
are in a directory called dcuser, which sits inside a
directory called home which sits inside the very top
directory in the hierarchy. The very top of the hierarchy is a directory
called / which is usually referred to as the root
directory. So, to summarize: dcuser is a directory in
home which is a directory in /.
Now enter the following command:
This jumps forward multiple levels to the .hidden
directory. Now go back to the home directory.
And then
These two commands have the same effect, they both take us to the
.hidden directory. The first one uses the absolute path,
giving the full address from the home directory. The second uses a
relative path, giving only the address from the working directory. A
full path always starts with a /. A relative path does
not.
A relative path is like getting directions from someone on the street. They tell you to “go right at the stop sign, and then turn left on Main Street”. That works great if you’re standing there together, but not so well if you’re trying to tell someone how to get there from another country. A full path is like GPS coordinates. It tells you exactly where something is no matter where you are right now.
You can usually use either a full path or a relative path depending on what is most convenient. If we are in the home directory, it is more convenient to enter the relative path since it involves less typing.
Over time, it will become easier for you to keep a mental note of the structure of the directories that you are using and how to quickly navigate amongst them.
Exercise 2(Intermediate): Relative path resolution
Using the filesystem diagram below, if pwd displays
/Users/thing, what will ls ../backup
display?
../backup: No such file or directory2012-12-01 2013-01-08 2013-01-272012-12-01/ 2013-01-08/ 2013-01-27/original pnas_final pnas_sub
- No: there is a directory
backupin/Users. - No: this is the content of
Users/thing/backup, but with..we asked for one level further up. - No: see previous explanation. Also, we did not specify
-Fto display/at the end of the directory names. - Yes:
../backuprefers to/Users/backup.
The commands cd, and cd ~ are very useful
for quickly navigating back to your home directory. We will be using the
~ character in later lessons to specify our home
directory.
- The
/,~, and..characters represent important navigational shortcuts. - Hidden files and directories start with
.and can be viewed usingls -a. - Relative paths specify a location starting from the current location, while absolute paths specify a location from the root of the file system.
Content from Working with Files and Directories
Last updated on 2026-02-20 | Edit this page
Overview
Questions
- How can I view and search file contents?
- How can I create, copy and delete files and directories?
- How can I control who has permission to modify a file?
- How can I repeat recently used commands?
Objectives
- View, search within, copy, move, and rename files. Create new directories.
- Use wildcards (
*) to perform operations on multiple files. - Make a file read only
- Use the
historycommand to view and repeat recently used commands.
Working with Files
Our data set: FASTQ files
Now that we know how to navigate around our directory structure, lets
start working with our sequencing files. We did a sequencing experiment
and have two results files, which are stored in our data
directory.
Wildcards
Navigate to your data directory.
We are interested in looking at the FASTA files in this directory. We can list all files with the .fastq extension using the command:
OUTPUT
agalactiae_18RS21/Streptococcus_agalactiae_18RS21.gbk agalactiae_H36B/Streptococcus_agalactiae_H36B.gbkThe * character is a special type of character called a
wildcard, which can be used to represent any number of any type of
character. Thus, *.gbk matches every file that ends with
.gbk.
Command History
If you want to repeat a command that you’ve run recently, you can access previous commands using the up arrow on your keyboard to go back to the most recent command. Likewise, the down arrow takes you forward in the command history.
A few more useful shortcuts:
- Ctrl+C will cancel the command you are writing, and give you a fresh prompt.
- Ctrl+R will do a reverse-search through your command history. This is very useful.
-
Ctrl+L or the
clearcommand will clear your screen.
You can also review your recent commands with the
history command, by entering:
to see a numbered list of recent commands. You can reuse one of these commands directly by referring to the number of that command.
For example, if your history looked like this:
OUTPUT
479 ls *
480 ls /usr/bin/*.sh
481 ls *.gbkthen you could repeat command #481 by entering:
Type ! (exclamation point) and then the number of the
command from your history. You will be glad you learned this when you
need to re-run very complicated commands.
Examining Files
We now know how to switch directories, run programs, and look at the contents of directories, but how do we look at the contents of files?
One way to examine a file is to print out all of the contents using
the program cat.
$ cat filenamecat is a terrific program, but when the file is really
big (as the files we have), it can be annoying to use. The program,
less, is useful for this case. less opens the
file as read only, and lets you navigate through it. The navigation
commands are identical to the man program.
Enter the following command:
OUTPUT
Streptococcus_agalactiae_18RS21.fna Streptococcus_agalactiae_18RS21.gbkSome navigation commands in less
| key | action |
|---|---|
| Space | to go forward |
| b | to go backward |
| g | to go to the beginning |
| G | to go to the end |
| q | to quit |
less also gives you a way of searching through files.
Use the “/” key to begin a search. Enter the word you would like to
search for and press enter. The screen will jump to the
next location where that word is found.
Shortcut: If you hit “/” then “enter”,
less will repeat the previous search. less
searches from the current location and works its way forward. Note, if
you are at the end of the file and search for the sequence “CAA”,
less will not find it. You either need to go to the
beginning of the file (by typing g) and search again using
/ or you can use ? to search backwards in the
same way you used / previously.
For instance, let’s search forward for the sequence
TTTTT in our file. You can see that we go right to that
sequence, what it looks like, and where it is in the file. If you
continue to type / and hit return, you will move forward to
the next instance of this sequence motif. If you instead type
? and hit return, you will search backwards and move up the
file to previous examples of this motif.
Remember, the man program actually uses
less internally and therefore uses the same commands, so
you can search documentation using “/” as well!
There’s another way that we can look at files, and in this case, just look at part of them. This can be particularly useful if we just want to see the beginning or end of the file, or see how it’s formatted.
The commands are head and tail and they let
you look at the beginning and end of a file, respectively.
OUTPUT
LOCUS AAJO01000169.1 2501 bp DNA linear UNK
DEFINITION Streptococcus agalactiae 18RS21
ACCESSION AAJO01000169.1
KEYWORDS .
SOURCE Streptococcus agalactiae 18RS21.
ORGANISM Streptococcus agalactiae 18RS21
Bacteria; Terrabacteria group; Firmicutes; Bacilli;
Lactobacillales; Streptococcaceae; Streptococcus; Streptococcus
agalactiae.
FEATURES Location/QualifiersOUTPUT
241 gcagtatcct gagtacggcg agacacgcga aatctcgtcg gaatctggga ggaccatctc
301 ccaaccctaa atactctcta gtgaccgata gtgaaccagt accgtgaggg aaaggtgaaa
361 agcaccccgg gaggggagtg aaatagaacc tgaaaccgtg tgcctacaac aagttcgagc
421 ccgttaatgg gtgagagcgt gccttttgta gaatgaaccg gcgagttacg atatgatgcg
481 aggttaagtt gaagagacgg agccgtaggg aaaccgagtc ttaatagggc gtcatagtat
541 catgttgtag acccgaaacc atgtgaccta cccatgagca ggttgaaggt gaggtaaaac
601 tcactggagg accgaaccag ggcacgttga aaagtgcttg gatgacttgt gggtagcgga
661 gaaattcaaa cgaacttgga gatagctggt tctctccgaa atagctttag ggctagcgtc
721 gatgttaagt ctcttggagt aga
//The -n option to either of these commands can be used to
print the first or last n lines of a file.
OUTPUT
>LOCUS AAJO01000169.1 2501 bp DNA linear UNKOUTPUT
//Details on the GBK format
Although it looks complicated (and it is), it’s easy to understand the fastq format with a little decoding. Some rules about the format include…
| Line | Description |
|---|---|
| 1 | May start with a “;” or “>”, follows by a name and/or a unique identifier for the sequence, and may also contain additional information |
| 2 | The actual DNA sequence |
| 3 | If there are more sequences, it always begins with a ‘>’, and info like info in line 1 |
We can view the first complete read in one of the files our dataset
by using head to look at the first four lines.
OUTPUT
LOCUS AAJO01000169.1 2501 bp DNA linear UNK
DEFINITION Streptococcus agalactiae 18RS21
ACCESSION AAJO01000169.1
KEYWORDS .
SOURCE Streptococcus agalactiae 18RS21.
ORGANISM Streptococcus agalactiae 18RS21
Bacteria; Terrabacteria group; Firmicutes; Bacilli;
Lactobacillales; Streptococcaceae; Streptococcus; Streptococcus
agalactiae.
FEATURES Location/QualifiersCreating, moving, copying, and removing
Now we can move around in the file structure, look at files, and search files. But what if we want to copy files or move them around or get rid of them? Most of the time, you can do these sorts of file manipulations without the command line, but there will be some cases (like when you’re working with a remote computer like we are for this lesson) where it will be impossible. You’ll also find that you may be working with hundreds of files and want to do similar manipulations to all of those files. In cases like this, it’s much faster to do these operations at the command line.
Copying Files
When working with computational data, it’s important to keep a safe copy of that data that can’t be accidentally overwritten or deleted. For this lesson, our raw data is our GBK files. We don’t want to accidentally change the original files, so we’ll make a copy of them and change the file permissions so that we can read from, but not write tcdo, the files.
First, let’s make a copy of one of our GBK files using the
cp command.
Navigate to the ~/pan_workshop/data/agalactiae_18RS21
directory and enter:
OUTPUT
Streptococcus_agalactiae_18RS21_copy.gbk
Streptococcus_agalactiae_18RS21.fna
Streptococcus_agalactiae_18RS21.gbkWe now, for example, have two copies of the
Streptococcus_agalactiae_18RS21.gbk file, one of them named
Streptococcus_agalactiae_18RS21_copy.gbk. We’ll move this
file to a new directory called backup where we’ll store our
backup data files.
Creating Directories
The mkdir command is used to make a directory. Enter
mkdir followed by a space, then the directory name you want
to create.
Moving / Renaming
We can now move our backup file to this directory. We can move files
around using the command mv.
OUTPUT
Streptococcus_agalactiae_18RS21_copy.gbkThe mv command is also how you rename files. Let’s
rename this file to make it clear that this is a backup.
BASH
$ cd backup
$ mv Streptococcus_agalactiae_18RS21_copy.gbk Streptococcus_agalactiae_18RS21_backup.gbk
$ lsOUTPUT
Streptococcus_agalactiae_18RS21_backup.gbkRemoving
When we want to remove a file or a directory we use the
rm command. By default, rmwill not delete
directories. You can tell rm to delete a directory using
the -r (recursive) option.
Let’s delete the backup directory we just made.
This will delete not only the directory, but all files within the directory. If you have write-protected files in the directory, you will be asked whether you want to override your permission settings.
If we want to modifiy a file without all the permissions you’ll be asked if you want to override your file permissions. for example:
OUTPUT
rm: remove write-protected regular file 'example.fastq'? If you enter n (for no), the file will not be deleted.
If you enter y, you will delete the file. This gives us an
extra measure of security, as there is one more step between us and
deleting our data files.
Important: The rm command permanently
removes the file. Be careful with this command. It doesn’t just nicely
put the files in the Trash. They’re really gone.
Exercise 1(Intermediate): Make backup folder with write-protected permissions
Starting in the
/home/dcuser/pan_workshop/data/agalactiae_18RS21 directory,
do the following:
- Make sure that you have deleted your backup directory and all files it contains.
- Create a copy of each of your GBK files. (Note: You’ll need to do this individually for each of the two FASTQ files. We haven’t learned yet how to do this with a wildcard.)
- Use a wildcard to move all of your backup files to a new backup directory.
- Change the permissions on all of your backup files to be write-protected.
rm -r backupcp Streptococcus_agalactiae_18RS21.gbk Streptococcus_agalactiae_18RS21_copy.gbk-
mkdir backupandmv *_copy.gbk backup -
chmod -w backup/*-copy.gbk
It’s always a good idea to check your work withls -l backup. You should see something like:
OUTPUT
-r-r--r-- 1 dcuser dcuser 4776510 jun 6 2022 Streptococcus_agalactiae_18RS21.gbk- You can view file contents using
less,cat,headortail. - The commands
cp,mv, andmkdirare useful for manipulating existing files and creating new directories. - You can view file permissions using
ls -land change permissions usingchmod. - The
historycommand and the up arrow on your keyboard can be used to repeat recently used commands.
Content from Redirection
Last updated on 2026-02-20 | Edit this page
Overview
Questions
- How can I search within files?
- How can I combine existing commands to do new things?
Objectives
- Employ the
grepcommand to search for information within files. - Print the results of a command to a file.
- Construct command pipelines with two or more stages.
Searching files
We discussed in a previous episode how to search within a file using
less. We can also search within files without even opening
them, using grep. grep is a command-line
utility for searching plain-text files for lines matching a specific set
of characters (sometimes called a string) or a particular pattern (which
can be specified using something called regular expressions). We’re not
going to work with regular expressions in this lesson, and are instead
going to specify the strings we are searching for. Let’s give it a
try!
Nucleotide abbreviations
The four nucleotides that appear in DNA are abbreviated
A, C, T and G.
Unknown nucleotides are represented with the letter N. An
N appearing in a sequencing file represents a position
where the sequencing machine was not able to confidently determine the
nucleotide in that position. You can think of an N as being
aNy nucleotide at that position in the DNA sequence.
We’ll search for strings inside of our fastq files. Let’s first make sure we are in the correct directory.
OUTPUT
JC1A_R1.fastq JC1A_R2.fastq JP4D_R1.fastq JP4D_R2.fastqSuppose we want to see how many reads in our file have really bad segments containing 10 consecutive unknown nucleotides (Ns).
Determining quality
In this lesson, we’re going to be manually searching for strings of
Ns within our sequence results to illustrate some
principles of file searching. It can be really useful to do this type of
searching to get a feel for the quality of your sequencing results,
however, in your research you will most likely use a bioinformatics tool
that has a built-in program for filtering out low-quality reads. You’ll
learn how to use one such tool in a
later lesson.
Let’s search for the string NNNNNNNNNN in the JC1A_R2.fastq file.
This command returns a lot of output to the terminal. Every single line in the JC1A_R2.fastq file that contains at least 10 consecutive Ns is printed to the terminal, regardless of how long or short the file is. We may be interested not only in the actual sequence which contains this string, but in the name (or identifier) of that sequence. We discussed in a previous lesson that the identifier line immediately precedes the nucleotide sequence for each read in a FASTQ file. We may also want to inspect the quality scores associated with each of these reads. To get all of this information, we will return the line immediately before each match and the two lines immediately after each match.
We can use the -B argument for grep to return a specific
number of lines before each match. The -A argument returns
a specific number of lines after each matching line. Here we want the
line before and the two lines after each matching
line, so we add -B1 -A2 to our grep command.
One of the sets of lines returned by this command is:
OUTPUT
@MISEQ-LAB244-W7:91:000000000-A5C7L:1:2111:5300:24013 2:N:0:TCGAAG
NNNNNNNNNNNCNANNANNNNNCGCCGGTGTTCTNCTGGGGNACGGANACCGAGTAGATCGGAACAGCGTCGTGGAGNGAAAGAGTGTAGATCCCGGTGGGCGGCGTATCATTAAAAAAAAAACCTGCTGGTCCTTGTCTC
+
AAA11BB3333BGG1GGEC1E?0E0B0BFDGFHD2FBH110A1BEE?A/BAFBDGH///>FEGGG><@/#//?#?/#//????########################################################################################################################################################################Exercise 1(Begginer): Using grep
Search for the sequence
GATCGAGAGGGGATAGGCGin theJC1A_R2.fastqfile. Have your search return all matching lines and the name (or identifier) for each sequence that contains a match.Search for the sequence
AAGTTin all FASTQ files. Have your search return all matching lines and the name (or identifier) for each sequence that contains a match.
grep -B1 GATCGAGAGGGGATAGGCG JC1A_R2.fastqgrep -B1 AAGTT *.fastq
Redirecting output
grep allowed us to identify sequences in our FASTQ files
that match a particular pattern. All of these sequences were printed to
our terminal screen, but in order to work with these sequences and
perform other operations on them, we will need to capture that output in
some way.
We can do this with something called “redirection”. The idea is that we are taking what would ordinarily be printed to the terminal screen and redirecting it to another location. In our case, we want to print this information to a file so that we can look at it later and use other commands to analyze this data.
The command for redirecting output to a file is
>.
Let’s try out this command and copy all the records (including all
four lines of each record) in our FASTQ files that contain ‘NNNNNNNNNN’
to another file called bad_reads.txt.
File extensions
You might be confused about why we’re naming our output file with a
.txt extension. After all, it will be holding FASTQ
formatted data that we’re extracting from our FASTQ files. Won’t it also
be a FASTQ file? The answer is, yes - it will be a FASTQ file and it
would make sense to name it with a .fastq extension.
However, using a .fastq extension will lead us to problems
when we move to using wildcards later in this episode. We’ll point out
where this becomes important. For now, it’s good that you’re thinking
about file extensions!
The prompt should sit there a little bit, and then it should look
like nothing happened. But type ls. You should see a new
file called bad_reads.txt.
We can check the number of lines in our new file using a command
called wc. wc stands for word
count. This command counts the number of words, lines, and
characters in a file.
OUTPUT
402 489 50076 bad_reads.txtThis will tell us the number of lines, words and characters in the
file. If we want only the number of lines, we can use the
-l flag for lines.
OUTPUT
402 bad_reads.txtBecause we asked grep for all four lines of each FASTQ
record, we need to divide the output by four to get the number of
sequences that match our search pattern.
Exercise 2(Intermediate): Using
wc
How many sequences in JC1A_R2.fastq contain at least 3
consecutive Ns?
Exercise 2(Intermediate): Using
wc (continued)
596 bad_reads.txt
{: .output}
We might want to search multiple FASTQ files for sequences that match
our search pattern. However, we need to be careful, because each time we
use the > command to redirect output to a file, the new
output will replace the output that was already present in the file.
This is called “overwriting” and, just like you don’t want to overwrite
your video recording of your kid’s first birthday party, you also want
to avoid overwriting your data files.
OUTPUT
24 bad_reads.txtThe old bad_reads.txt that counts bad quality reads from
file JC1A_R2.fastq with 402 lines has been erased. Instead
a new bad_reads.txt that contain 24 lines from bad reads
from JC1A_R1.fastq has been created. We can avoid
overwriting our files by using the command >>.
>> is known as the “append redirect” and will append
new output to the end of a file, rather than overwriting it.
OUTPUT
402 bad_reads.txtOUTPUT
426 bad_reads.txtThe output of our second call to wc shows that we have
not overwritten our original data. The final number of 426 lines results
from the adition of 402 reads from JC1A_R2.fastq file + 24
reads from JC1A_R1.fastq file. We can also do this for more
files with a single line of code by using a wildcard.
OUTPUT
427 bad_reads.txtFile extensions - part 2
This is where we would have trouble if we were naming our output file
with a .fastq extension. If we already had a file called
bad_reads.fastq (from our previous grep
practice) and then ran the command above using a .fastq
extension instead of a .txt extension, grep
would give us a warning.
OUTPUT
grep: input file 'bad_reads.fastq' is also the outputgrep is letting you know that the output file
bad_reads.fastq is also included in your grep
call because it matches the *.fastq pattern. Be careful
with this as it can lead to some unintended results.
Since we might have multiple different criteria we want to search
for, creating a new output file each time has the potential to clutter
up our workspace. We also so far haven’t been interested in the actual
contents of those files, only in the number of reads that we’ve found.
We created the files to store the reads and then counted the lines in
the file to see how many reads matched our criteria. There’s a way to do
this, however, that doesn’t require us to create these intermediate
files - the pipe command (|).
This is probably not a key on your keyboard you use very much, so
let’s all take a minute to find that key. What | does is
take the output that is scrolling by on the terminal and uses that
output as input to another command. When our output was scrolling by, we
might have wished we could slow it down and look at it, like we can with
less. Well it turns out that we can! We can redirect our
output from our grep call through the less
command.
We can now see the output from our grep call within the
less interface. We can use the up and down arrows to scroll
through the output and use q to exit less.
Redirecting output is often not intuitive, and can take some time to get used to. Once you’re comfortable with redirection, however, you’ll be able to combine any number of commands to do all sorts of exciting things with your data!
None of the command line programs we’ve been learning do anything all that impressive on their own, but when you start chaining them together, you can do some really powerful things very efficiently.
Writing for loops
Loops are key to productivity improvements through automation as they allow us to execute commands repeatedly. Similar to wildcards and tab completion, using loops also reduces the amount of typing (and typing mistakes). Loops are helpful when performing operations on groups of sequencing files, such as unzipping or trimming multiple files. We will use loops for these purposes in subsequent analyses, but will cover the basics of them for now.
When the shell sees the keyword for, it knows to repeat
a command (or group of commands) once for each item in a list. Each time
the loop runs (called an iteration), an item in the list is assigned in
sequence to the variable, and the commands inside the
loop are executed, before moving on to the next item in the list. Inside
the loop, we call for the variable’s value by putting $ in
front of it. The $ tells the shell interpreter to treat the
variable as a variable name and substitute its value in
its place, rather than treat it as text or an external command. In shell
programming, this is usually called “expanding” the variable.
Let’s write a for loop to show us the first two lines of the fastq
files we downloaded earlier. You will notice shell prompt changes from
$ to > and back again as we were typing in
our loop. The second prompt, >, is different to remind
us that we haven’t finished typing a complete command yet. A semicolon,
;, can be used to separate two commands written on a single
line.
The for loop begins with the formula
for <variable> in <group to iterate over>. In
this case, the word filename is designated as the variable
to be used over each iteration. In our case JC1A_R1.fastq
and JC1A_R2.fastq will be substituted for
filename because they fit the pattern of ending with .fastq
in directory we’ve specified. The next line of the for loop is
do. The next line is the code that we want to execute. We
are telling the loop to print the first two lines of each variable we
iterate over and save the information to a file. Finally, the word
done ends the loop.
Note that we are using >> to append the text to
our seq_info.txt file. If we used >, the
seq_info.txt file would be rewritten every time the loop
iterates, so it would only have text from the last variable used.
Instead, >> adds to the end of the file.
Using Basename in for loops
Basename is a function in UNIX that is helpful for removing a uniform
part of a name from a list of files. In this case, we will use basename
to remove the .fastq extension from the files that we’ve
been working with.
We see that this returns just the SRR accession, and no longer has the .fastq file extension on it.
OUTPUT
JC1A_R2If we try the same thing but use .fasta as the file
extension instead, nothing happens. This is because basename only works
when it exactly matches a string in the file.
OUTPUT
JC1A_R2.fastqBasename is really powerful when used in a for loop. It allows to access just the file prefix, which you can use to name things. Let’s try this.
Inside our for loop, we create a new name variable. We call the
basename function inside the parenthesis, then give our variable name
from the for loop, in this case ${filename}, and finally
state that .fastq should be removed from the file name.
It’s important to note that we’re not changing the actual files, we’re
creating a new variable called name. The line > echo $name will print
to the terminal the variable name each time the for loop runs. Because
we are iterating over two files, we expect to see two lines of
output.
Exercise 3(Advanced): Using
basename
Print the file prefix of all of the .txt files in our
current directory.
One way this is really useful is to move files. Let’s rename all of
our .txt files using mv so that they have the years on
them, which will document when we created them.
BASH
$ for filename in *.txt
> do
> name=$(basename ${filename} .txt)
> mv ${filename} ${name}_2019.txt
> done-
grepis a powerful search tool with many options for customization. -
>,>>, and|are different ways of redirecting output. -
command > fileredirects a command’s output to a file. -
command >> fileredirects a command’s output to a file without overwriting the existing contents of the file. -
command_1 | command_2redirects the output of the first command as input to the second command. - for loops are used for iteration
-
basenamegets rid of repetitive parts of names
Content from Writing Scripts and Working with Data
Last updated on 2026-02-20 | Edit this page
Overview
Questions
- How can we automate a commonly used set of commands?
Objectives
- Use the
nanotext editor to modify text files. - Write a basic shell script.
- Use the
bashcommand to execute a shell script. - Use
chmodto make a script an executable program.
Writing files
We’ve been able to do a lot of work with files that already exist, but what if we want to write our own files. We’re not going to type in a FASTA file, but we’ll see as we go through other tutorials, there are a lot of reasons we’ll want to write a file, or edit an existing file.
To add text to files, we’re going to use a text editor called Nano.
We’re going to create a file to take notes about what we’ve been doing
with the data files in
~/dc_workshopd/data/untrimmed_fastq.
This is good practice when working in bioinformatics. We can create a
file called a README.txt that describes the data files in
the directory or documents how the files in that directory were
generated. As the name suggests it’s a file that we or others should
read to understand the information in that directory.
Let’s change our working directory to
~/dc_workshop/data/untrimmed_fastq using cd,
then run nano to create a file called
README.txt:
You should see something like this:
The text at the bottom of the screen shows the keyboard shortcuts for
performing various tasks in nano. We will talk more about
how to interpret this information soon.
Which Editor?
When we say, “nano is a text editor,” we really do mean
“text”: it can only work with plain character data, not tables, images,
or any other human-friendly media. We use it in examples because it is
one of the least complex text editors. However, because of this trait,
it may not be powerful enough or flexible enough for the work you need
to do after this workshop. On Unix systems (such as Linux and Mac OS X),
many programmers use Emacs or Vim (both of which require more time to
learn), or a graphical editor such as Gedit. On Windows, you may
wish to use Notepad++.
Windows also has a built-in editor called notepad that can
be run from the command line in the same way as nano for
the purposes of this lesson.
No matter what editor you use, you will need to know where it searches for and saves files. If you start it from the shell, it will (probably) use your current working directory as its default location. If you use your computer’s start menu, it may want to save files in your desktop or documents directory instead. You can change this by navigating to another directory the first time you “Save As…”
Let’s type in a few lines of text. Describe what the files in this
directory are or what you’ve been doing with them. Once we’re happy with
our text, we can press Ctrl-O (press the
Ctrl or Control key and, while holding it down,
press the O key) to write our data to disk. You’ll be asked
what file we want to save this to: press Return to accept the
suggested default of README.txt.
Once our file is saved, we can use Ctrl-X to quit the editor and return to the shell.
Control, Ctrl, or ^ Key
The Control key is also called the “Ctrl” key. There are various ways in which using the Control key may be described. For example, you may see an instruction to press the Ctrl key and, while holding it down, press the X key, described as any of:
Control-XControl+XCtrl-XCtrl+X^XC-x
In nano, along the bottom of the screen you’ll see
^G Get Help ^O WriteOut. This means that you can use
Ctrl-G to get help and
Ctrl-O to save your file.
Now you’ve written a file. You can take a look at it with
less or cat, or open it up again and edit it
with nano.
Exercise 1(Begginer): Edit a file with nano
Open README.txt and add the date to the top of the file
and save the file.
Writing scripts
A really powerful thing about the command line is that you can write scripts. Scripts let you save commands to run them and also lets you put multiple commands together. Though writing scripts may require an additional time investment initially, this can save you time as you run them repeatedly. Scripts can also address the challenge of reproducibility: if you need to repeat an analysis, you retain a record of your command history within the script.
One thing we will commonly want to do with sequencing results is pull out bad reads and write them to a file to see if we can figure out what’s going on with them. We’re going to look for reads with long sequences of N’s like we did before, but now we’re going to write a script, so we can run it each time we get new sequences, rather than type the code in by hand each time.
Bad reads have a lot of N’s, so we’re going to look for
NNNNNNNNNN with grep. We want the whole FASTQ
record, so we’re also going to get the one line above the sequence and
the two lines below. We also want to look in all the files that end with
.fastq, so we’re going to use the *
wildcard.
We’re going to create a new file to put this command in. We’ll call
it bad-reads-script.sh. The sh isn’t required,
but using that extension tells us that it’s a shell script.
Type your grep command into the file and save it as
before. Be careful that you did not add the $ at the
beginning of the line.
Now comes the neat part. We can run this script. Type:
It will look like nothing happened, but now if you look at
scripted_bad_reads.txt, you can see that there are now
reads in the file.
Exercise 2(Intermediate): Edit a script
We want the script to tell us when it’s done.
Versioning scripts with Git and GitHub
A good coding practice is to keep versions of the scripts you code. That way, you can control what it is functional on each version, and you can always come back to older versions if something goes wrong with the new ones. Git is a version control system that helps you keep track of the entire history of the scripts that you are working on and facilitates collaboration on projects. GitHub is a web-based service for version control and online collaboration. It is a hosting service for Git repositories, and is a handy platform for backup of software code and files. GitHub acts as a social networking site for software developers where they can manage projects and build their portfolio.
Git is alradey installed in you machine, let’s have a look at its help command:
The first lines of the git help are displayed below:
OUTPUT
usage: git [--version] [--help] [-C <path>] [-c name=value]
[--exec-path[=<path>]] [--html-path] [--man-path] [--info-path]
[-p | --paginate | --no-pager] [--no-replace-objects] [--bare]
[--git-dir=<path>] [--work-tree=<path>] [--namespace=<name>]
<command> [<args>]
These are common Git commands used in various situations:
start a working area (see also: git help tutorial)
clone Clone a repository into a new directory To learn more about Git and GitHub platforms for version control and collaborative development, check the Software Carpentries website https://swcarpentry.github.io/git-novice/
Making the script into a program
We had to type bash because we needed to tell the
computer what program to use to run this script. Instead we can turn
this script into its own program. We need to tell it that it’s a program
by making it executable. We can do this by changing the file
permissions. We talked about permissions in an
earlier episode.
First, let’s look at the current permissions.
OUTPUT
-rw-rw-r-- 1 dcuser dcuser 0 Oct 25 21:46 bad-reads-script.shWe see that it says -rw-r--r--. This shows that the file
can be read by any user and written to by the file owner (you). We want
to change these permissions so that the file can be executed as a
program. We use the command chmod like we did earlier when
we removed write permissions. Here we are adding (+)
executable permissions (+x).
Now let’s look at the permissions again.
OUTPUT
-rwxrwxr-x 1 dcuser dcuser 0 Oct 25 21:46 bad-reads-script.shNow we see that it says -rwxr-xr-x. The x’s
that are there now tell us we can run it as a program. So, let’s try it!
We’ll need to put ./ at the beginning so the computer knows
to look here in this directory for the program.
The script should run the same way as before, but now we’ve created our very own computer program!
You will learn more about writing scripts in a later lesson.
It is good practice to keep any large files compressed while you are
not using them. In this way you save storage space, you will see that
you will appreciate it when you advance in your analysis. So, since we
will not use the FASTQ files for now, let’s compress them. And run
ls -lh to confirm that they are compressed.
BASH
$ gzip ~/dc_workshop/data/untrimmed_fastq/*.fastq
$ ls -lh ~/dc_workshop/data/untrimmed_fastq/*.fastq.gzOUTPUT
total 428M
-rw-r--r-- 1 dcuser dcuser 24M Nov 26 12:36 JC1A_R1.fastq.gz
-rw-r--r-- 1 dcuser dcuser 24M Nov 26 12:37 JC1A_R2.fastq.gz
-rw-r--r-- 1 dcuser dcuser 179M Nov 26 12:44 JP4D_R1.fastq.gz
-rw-r--r-- 1 dcuser dcuser 203M Nov 26 12:51 JP4D_R2.fastq.gzMoving and downloading data
So far, we’ve worked with data that is pre-loaded on the instance in the cloud. Usually, however, most analyses begin with moving data onto the instance. Below we’ll show you some commands to download data onto your instance, or to move data between your computer and the cloud.
Getting data from the cloud
There are two programs that will download data from a remote server
to your local (or remote) machine: wget and
curl. They were designed to do slightly different tasks by
default, so you’ll need to give the programs somewhat different options
to get the same behaviour, but they are mostly interchangeable.
wgetis short for “world wide web get”, and it’s basic function is to download web pages or data at a web address.cURLis a pun, it is suppose to be read as “see URL”, so it’s basic function is to display webpages or data at a web address.
Which one you need to use mostly depends on your operating system, as most computers will only have one or the other installed by default.
Let’s say you want to download some data from Ensembl. We’re going to
download a very small tab-delimited file that just tells us what data is
available on the Ensembl bacteria server. Before we can start our
download, we need to know whether we’re using curl or
wget.
To see which program you have type:
which is a BASH program that looks through everything
you have installed, and tells you what folder it is installed to. If it
can’t find the program you asked for, it returns nothing, i.e. gives you
no results.
On Mac OSX, you’ll likely get the following output:
OUTPUT
/usr/bin/curlOUTPUT
$This output means that you have curl installed, but not
wget.
Once you know whether you have curl or wget
use one of the following commands to download the file:
or
BASH
$ cd
$ curl -O ftp://ftp.ensemblgenomes.org/pub/release-37/bacteria/species_EnsemblBacteria.txtSince we wanted to download the file rather than just view
it, we used wget without any modifiers. With
curl however, we had to use the -O flag, which
simultaneously tells curl to download the page instead of
showing it to us and specifies that it should save the
file using the same name it had on the server:
species_EnsemblBacteria.txt
It’s important to note that both curl and
wget download to the computer that the command line belongs
to. So, if you are logged into AWS on the command line and execute the
curl command above in the AWS terminal, the file will be
downloaded to your AWS machine, not your local one.
Moving files between your laptop and your instance
What if the data you need is on your local computer, but you need to get it into the cloud? There are also several ways to do this, but it’s always easier to start the transfer locally. This means if you’re typing into a terminal, the terminal should not be logged into your instance, it should be showing your local computer. If you’re using a transfer program, it needs to be installed on your local machine, not your instance.
Transferring data between your local machine and the cloud
Uploading data to your virtual machine with scp
scp stands for ‘secure copy protocol’, and is a widely
used UNIX tool for moving files between computers. The simplest way to
use scp is to run it in your local terminal, and use it to
copy a single file:
Note that you are always running scp locally, but that
doesn’t mean that you can only move files from your local
computer. A command like:
To move it back, you just re-order the to and from fields:
Uploading data to your virtual machine with scp
- Open the terminal and use the
scpcommand to upload a file (e.g. local_file.txt) to the dcuser home directory:
Downloading data from your virtual machine with
scp
Let’s download a text file from our remote machine. You should have a file that contains bad reads called ~/data/untrimmed_fastq/scripted_bad_reads.txt.
Tip: If you are looking for another (or any really) text file in your home directory to use instead try
- Download the bad reads file in ~/data/scripted_bad_reads.txt to your home ~/Download directory using the following command (make sure you use substitute dcuser@ ip.address with your remote login credentials):
BASH
$ scp dcuser@ip.address:/home/dcuser/dc_workshop/data/untrimmed_fastq/scripted_bad_reads.txt. ~/Downloads- Scripts are a collection of commands executed together.
- Transferring information to and from virtual and local computers.
Content from Project Organization
Last updated on 2026-02-20 | Edit this page
Overview
Questions
- How can I organize my file system for a new bioinformatics project?
- How can I document my work?
Objectives
- Create a file system for a bioinformatics project.
- Explain what types of files should go in your
docs,data, andresultsdirectories. - Use the
historycommand and a text editor likenanoto document your work on your project.
Getting your project started
Project organization is one of the most important parts of a sequencing project, and yet is often overlooked amidst the excitement of getting a first look at new data. Of course, while it is best to get yourself organized before you even begin your analyses, it is never too late to start, either.
You should approach your sequencing project similarly to how you do a biological experiment and this ideally begins with experimental design. We’re going to assume that you’ve already designed a beautiful sequencing experiment to address your biological question, collected appropriate samples, and that you have enough statistical power to answer the questions you’re interested in asking. These steps are all incredibly important, but beyond the scope of our course. For all of those steps (collecting specimens, extracting DNA, prepping your samples) you’ve likely kept a lab notebook that details how and why you did each step. However, the process of documentation doesn’t stop at the sequencer!
Genomics projects can quickly accumulate hundreds of files across tens of folders. Every computational analysis you perform over the course of your project is going to create many files, which can especially become a problem when you’ll inevitably want to run some of those analyses again. For instance, you might have made significant headway into your project, but then have to remember the PCR conditions you used to create your sequencing library months prior.
Other questions might arise along the way:
- What were your best alignment results?
- Which folder were they in: Analysis1, AnalysisRedone, or AnalysisRedone2?
- Which quality cutoff did you use?
- What version of a given program did you implement your analysis in?
Good documentation is key to avoiding this issue, and luckily enough, recording your computational experiments is even easier than recording lab data. Copy/Paste will become your best friend, sensible file names will make your analysis understandable by you and your collaborators, and writing the methods section for your next paper will be easy! Remember that in any given project of yours, it’s worthwhile to consider a future version of yourself as an entirely separate collaborator. The better your documenation is, the more this ‘collaborator’ will feel indebted to you!
With this in mind, let’s have a look at the best practices for documenting your genomics project. Your future self will thank you.
In this exercise we will setup a file system for the project we will be working on during this workshop.
We will start by creating a directory that we can use for the rest of
the workshop. First navigate to your home directory. Then confirm that
you are in the correct directory using the pwd command.
You should see the output:
OUTPUT
/home/dcuser Tip
If you aren’t in your home directory, the easiest way to get there is
to enter the command cd, which always returns you to
home.
Exercise 1(Begginer): Making and organized file system
Use the mkdir command to make the following
directories:
workshopworkshop/docsworkshop/dataworkshop/results
Use ls -R to verify that you have created these
directories. The -R option for ls stands for
recursive. This option causes ls to return the contents of
each subdirectory within the directory iteratively.
You should see the following output:
OUTPUT
workshop/:
data docs results
workshop/data:
workshop/docs:
workshop/results: Organizing your files
Before beginning any analysis, it’s important to save a copy of your raw data. The raw data should never be changed. Regardless of how sure you are that you want to carry out a particular data cleaning step, there’s always the chance that you’ll change your mind later or that there will be an error in carrying out the data cleaning and you’ll need to go back a step in the process. Having a raw copy of your data that you never modify guarantees that you will always be able to start over if something goes wrong with your analysis. When starting any analysis, you can make a copy of your raw data file and do your manipulations on that file, rather than the raw version. We learned in a previous episode how to prevent overwriting our raw data files by setting restrictive file permissions.
You can store any results that are generated from your analysis in
the results folder. This guarantees that you won’t confuse
results file and data files in six months or two years when you are
looking back through your files in preparation for publishing your
study.
The docs folder is the place to store any written
analysis of your results, notes about how your analyses were carried
out, and documents related to your eventual publication.
Documenting your activity on the project
When carrying out wet-lab analyses, most scientists work from a written protocol and keep a hard copy of written notes in their lab notebook, including any things they did differently from the written protocol. This detailed record-keeping process is just as important when doing computational analyses. Luckily, it’s even easier to record the steps you’ve carried out computational than it is when working at the bench.
The history command is a convenient way to document all
the commands you have used while analyzing and manipulating your project
files. Let’s document the work we have done on our project so far.
View the commands that you have used so far during this session using
history:
The history likely contains many more commands than you have used for the current project. Let’s view the last several commands that focus on just what we need for this project.
View the last n lines of your history (where n = approximately the last few lines you think relevant). For our example, we will use the last 7:
Exercise 2(Intermediate): Creating a record of the used commands
Using your knowledge of the shell, use the append redirect
>> to create a file called
workshop_log_XXXX_XX_XX.sh (Use the four-digit year,
two-digit month, and two digit day, e.g.
workshop_log_2021_03_25.sh)
You may have noticed that your history contains the
history command itself. To remove this redundancy from our
log, let’s use the nano text editor to fix the file:
(Remember to replace the 2021_03_25 with your workshop
date.)
From the nano screen, you can use your cursor to
navigate, type, and delete any redundant lines.
Add a date line and comment to the line where you have created the directory, for example:
bash treats the # character as a comment
character. Any text on a line after a # is ignored by bash
when evaluating the text as code.
Next, remove any lines of the history that are not relevant by
navigating to those lines and using your delete key. Save your file and
close nano.
Your file should look something like this:
OUTPUT
# 2021_03_25
# Created sample directories for the Data Carpentry workshop
mkdir workshop
mkdir workshop/docs
mkdir workshop/data
mkdir workshop/resultsIf you keep this file up to date, you can use it to re-do your work
on your project if something happens to your results files. To
demonstrate how this works, first delete your workshop
directory and all of its subdirectories. Look at your directory contents
to verify the directory is gone.
OUTPUT
dc_workshop R workshop workshop_log_2021_03_25.sh Then run your workshop log file as a bash script. You should see the
workshop directory and all of its subdirectories
reappear.
OUTPUT
workshop workshop_log_2021_03_25.txtIt’s important that we keep our workshop log file outside of our
workshop directory if we want to use it to recreate our
work. It’s also important for us to keep it up to date by regularly
updating with the commands that we used to generate our results
files.
Congratulations! You’ve finished your introduction to using the shell for metagenomics projects. You now know how to navigate your file system, create, copy, move, and remove files and directories, and automate repetitive tasks using scripts and wildcards. With this solid foundation, you’re ready to move on to apply all of these new skills to carrying out more sophisticated bioinformatics analysis work. Don’t worry if everything doesn’t feel perfectly comfortable yet. We’re going to have many more opportunities for practice as we move forward on our bioinformatics journey!
References
A Quick Guide to Organizing Computational Biology Projects
- Spend the time to organize your file system when you start a new project. Your future self will thank you!
- Always save a write-protected copy of your raw data.